Search
This report summarises the epidemiology of hospitalisations with laboratory-confirmed influenza during the 2019 influenza season. The Influenza Complications Alert Network (FluCAN) is a sentinel hospital-based surveillance program that operates at sites in all jurisdictions in Australia.
Healthcare personnel (HCP) are a priority group for annual influenza vaccination. Few studies have assessed the validity of recall of prior influenza vaccination status among HCP, especially for more than one preceding season.
Pregnant women are recognised as being at risk of serious illness from influenza. Despite this, and longstanding national recommendations for vaccination in pregnancy, vaccine uptake remains suboptimal. This study aims to determine factors associated with women declining influenza vaccination in pregnancy.
Group A β-haemolytic Streptococcus (GAS), a Gram-positive bacterium, causes skin, mucosal and systemic infections. Repeated GAS infections can lead to autoimmune diseases acute rheumatic fever (ARF) and rheumatic heart disease (RHD). Aboriginal and Torres Strait Islander peoples in Australia have the highest rates of ARF and RHD in the world.
The relationship between diabetic ketoacidosis (DKA) at diagnosis of type 1 diabetes and long-term glycemic control varies between studies. We aimed, firstly, to characterize the association of DKA and its severity with long-term HbA1c in a large contemporary cohort, and secondly, to identify other independent determinants of long-term HbA1c.
Polymicrobial sepsis is associated with worse patient outcomes than monomicrobial sepsis. Routinely used culture-dependent microbiological diagnostic techniques have low sensitivity, often leading to missed identification of all causative organisms.
Since the discovery of MECP2 duplication syndrome (MDS) in 1999, efforts to characterise this disorder have been limited by a lack of large datasets, with small case series often favouring the reporting of certain conditions over others. This study is the largest to date, featuring 134 males and 20 females, ascertained from the international MECP2 Duplication Database (MDBase).
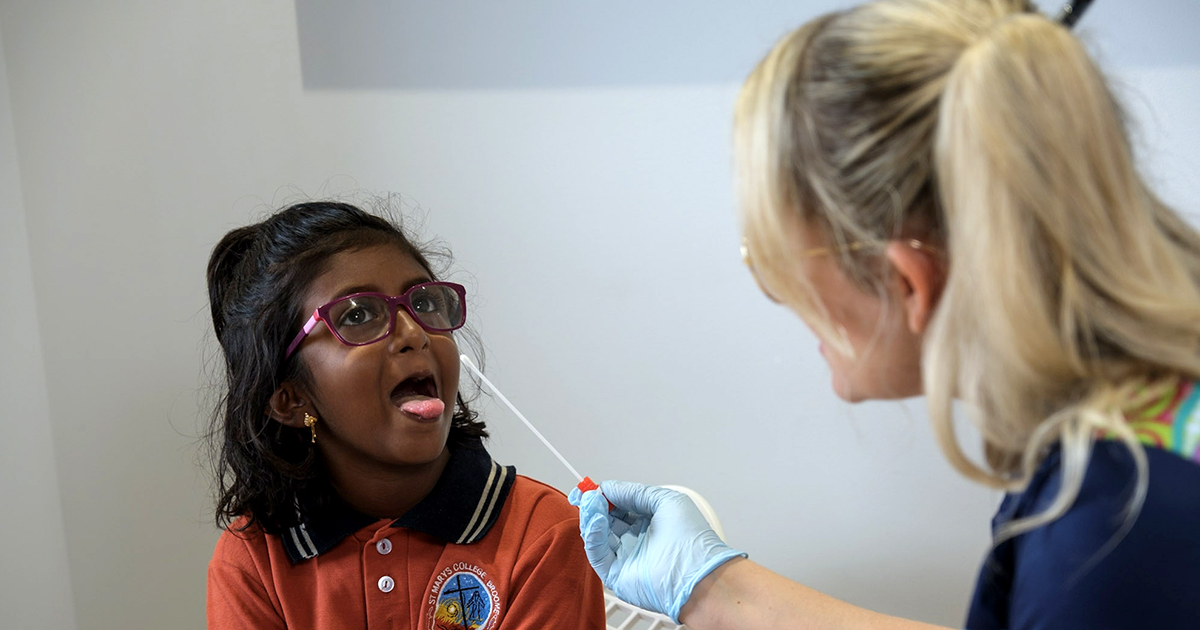
New research has revealed that molecular point-of-care testing (POCT) can significantly improve detection of Group A Streptococcal (Strep A) pharyngitis compared to laboratory culture – particularly in remote communities, where results can take up to seven days – unlocking a major opportunity to prevent rheumatic

Congratulations to Alec Truswell, Tamara Veselinović, Anastasia Phillips, and Wenna Lee for being awarded Seed Funding through the Wesfarmers Centre of Vaccines & Infectious Diseases, each valued at up to $25,000, and to Sonia McAlister, the recipient of the 2026 Riley Hughes Memorial Award in Pertussis Research.

Researchers at The Kids Research Institute Australia hope an Australian-first nasal spray vaccine for two to 11-year-olds will boost flu vaccination this winter.
