Search
Read about Professor Tim Jones and Associate Professor Liz Davis, co-directors of the Children's Diabetes Centre.
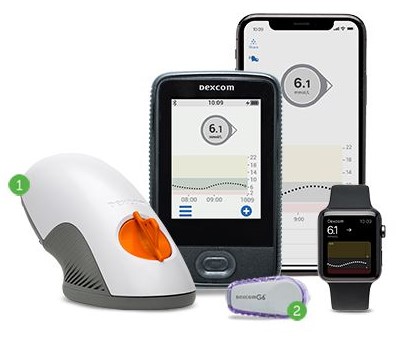
A new Q&A series focusing on the different research themes within the Children’s Diabetes Centre - technology.
The Rio Tinto Children’s Diabetes Centre; a Breakthrough T1D Centre of Excellence at The Kids Research Institute Australia and Perth Children’s Hospital (PCH), is a global hub for research into type 1 diabetes (T1D) in children.

What support do families with early-stage T1D need? Screening for the risk of developing T1D is now an option. But as exciting as screening is, we

In early 2024, Dr Aveni Haynes and her team were awarded Telethon Trust funding for their project Supporting Families.

Researcher Spotlight - Dr Aveni Haynes

Congratulations to Principal Research Fellow Dr Aveni Haynes from The Kids Research Institute Australia and the University of Western Australia.

Fantastic news for two of our talented researchers whose papers were featured in the Advanced Technologies & Treatments for Diabetes (ATTD) 2024 Yearbook.

Proudly funded by a Telethon Trust Research Grant, the Supporting Families project started in 2024 with the aim of co-designing a new clinical pathway for children with early stage type 1 diabetes (T1D).
The National Institute for Clinical Excellence updated guidance for continuous glucose monitoring (CGM) in 2022, recommending that CGM be available to all people living with type 1 diabetes. Manufacturers can trade in the UK with Conformité Européenne (CE) marking without an initial national assessment. The regulatory process for CGM CE marking, in contrast to the Food and Drug Administration (FDA) and Australian Therapeutic Goods Administration (TGA) process, is described.
