Search
The State Government has launched a new strategy for improving immunisation rates among WA children, to prevent outbreaks of diseases like measles & meningitis

Hundreds of healthy volunteers are needed in Perth to test the effectiveness of a current 'flu vaccine.
Just under 150 Perth women have contributed to major international research at the Telethon Institute for Child Health Research.
Perth researchers have begun a trial to test the effectiveness of a new vaccine to protect against the potentially deadly bird flu.
The Telethon Institute for Child Health Research today announced a new study investigating a vaccine to prevent infection of Human Papilloma Virus (HPV).

Clinical Research Manager

Head, Infectious Disease Implementation Research

Co-head, Bacterial Respiratory Infectious Disease Group (BRIDG)
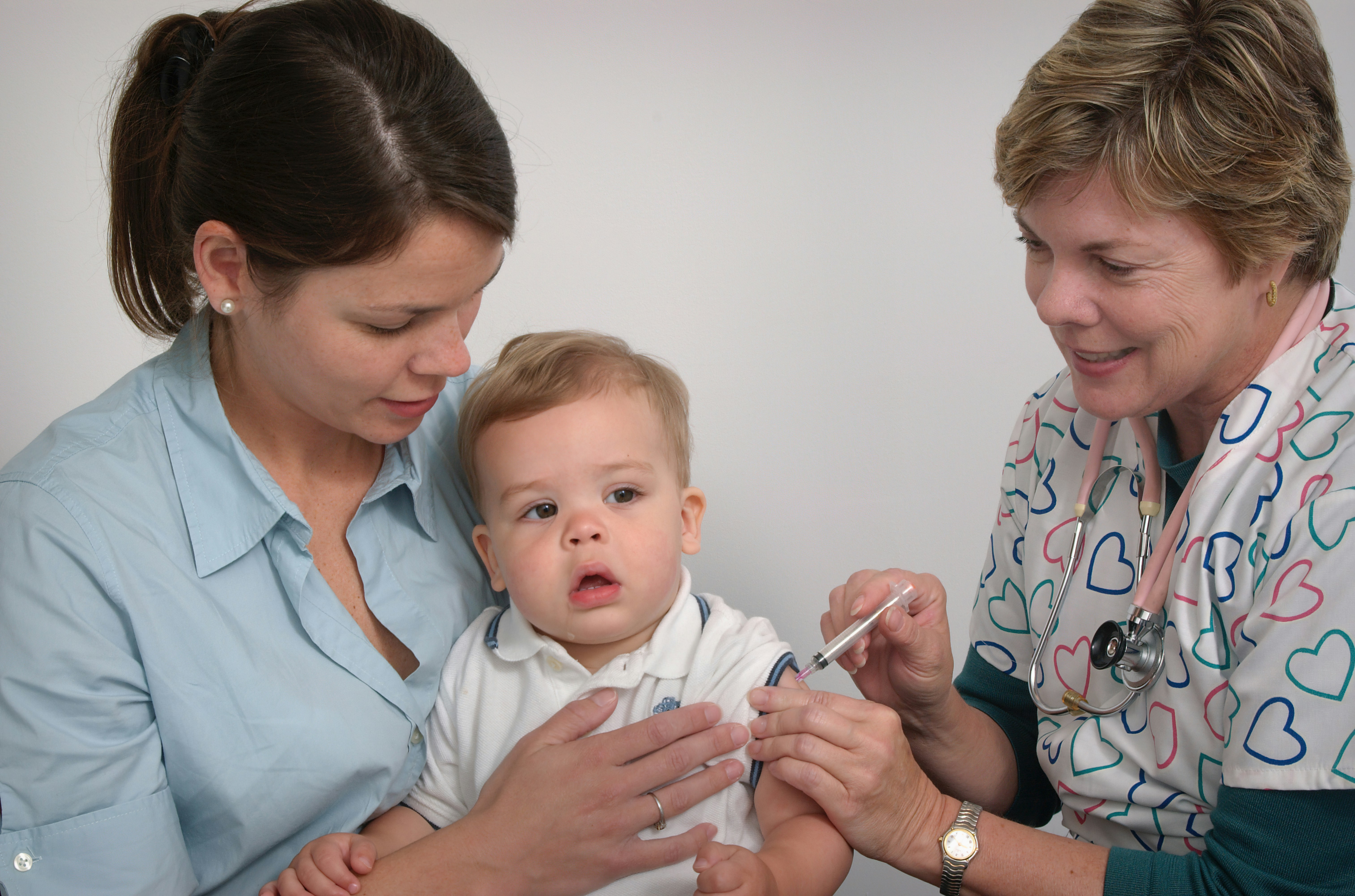
Researchers at The Kids Research Institute Australia have helped map the global impact of life saving vaccines to mark the 50-year anniversary of the Expanded Programme on Immunisation (EPI).

World-first immunisations providing protection against deadly respiratory syncytial virus (RSV) could be just months away thanks to global research efforts spanning multiple decades.
